Abstract
INTRODUCTION Chronic active Epstein-Barr virus (CAEBV) disease is a high-mortality disease, which is characterized by persistent infectious mononucleosis-like symptoms. There is no standard treatment of CAEBV and allogeneic hematopoietic stem cell transplantation (HSCT) was considered as an only potentially therapeutic approach. PD-1 inhibitors have achieved high response in many Epstein-Barr virus-related diseases. In this single-center retrospective analysis, we report the outcomes of PD-1 inhibitors treatment of CAEBV.
METHODS Sixteen CAEBV patients were treated with PD-1 inhibitors between 6/1/2017 and 12/31/2021. Hemophagocytic lymphohistiocytosis (HLH) was excluded. Patients received Nivolumab or Sindilizumab therapy every 3 to 4 weeks, 100 to 200 mg intravenously. Clinical and laboratory assessments were performed on patients before each infusion during treatment. Adverse events were evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 5.0.
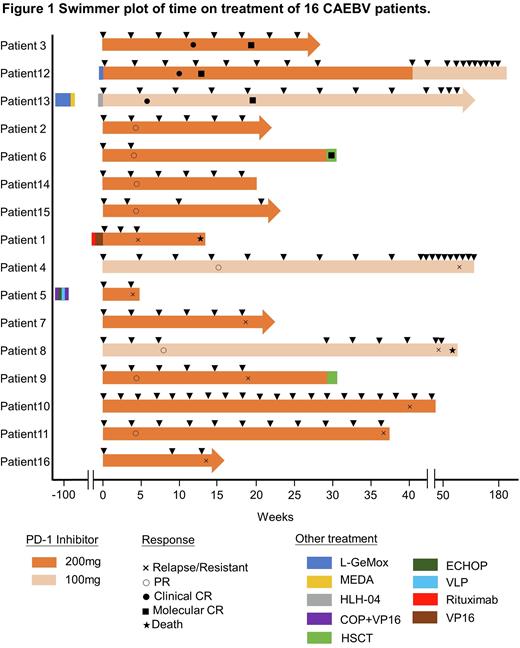
RESULTS Among the sixteen patients with a median age at onset of 33 years (range, 11-67 years), twelve patients responded to PD-1 inhibitors, and their median duration of relapse-free survival (RFS) was 10.3 months (range, 1.0-54.8 months). Three of them achieved clinical complete response (clinical CR), as well as molecular CR, and nine patients achieved partial response (PR) (Figure 1). Four of the nine PR patients converted from PR to no response (NR), and five remained in PR. The remaining four patients did not respond to PD-1 inhibitor therapy. One patient eventually died from a severe lung infection and one patient died of severe liver failure. The median time and cycles from the first application of PD-1 inhibitor to clinical CR in three patients with clinical CR were 43 days (range, 30-72 days) and 3 cycles (range, 2-4 cycles).
Patient 14 suffered immune-related pancreatitis (Grade 2, CTCAE 5.0) after 6 cycles of PD-1 inhibitor therapy. Four patients developed persistent or intermittent high fever after the first infusion of PD-1 inhibitors. Other than that, no treatment-related adverse events have been observed.
There was no correlation of treatment outcome with blood count, aspartate aminotransferase (AST) level, alanine aminotransferase (ALT) level, LDH level, cytokine level or ferritin level. Comparing the NK cell function of responders and non-responders, the test results showed higher numbers of patients with defective NK cell function among non-responders. The immunohistochemical analysis showed that patients with high PD-L1 expression in tumor tissue may have a more significant response to PD-1 inhibitors.
Genetic testing indicated that, except for 1 patient without gene sequencing and 8 patients with no related gene mutation detected, the other 7 patients were detected with potentially pathogenic or susceptibility mutations.
CONCLUSION In patients with CAEBV, PD-1 inhibitors have tolerable toxicity and comparable outcomes while improving quality of life and financial toxicity. Larger prospective studies and longer follow-up time is needed to be conducted.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.